calculate the solubility of mg oh 2 in water
17.4 Solvability and pH
Scholarship Objective
- To translate why the solvability of many compounds depends on pH.
The solubility of many compounds depends strongly along the pH of the solution. For example, the anion in many sparingly meltable salts is the conjugate base of a diluted acid that English hawthorn get over protonated in solution. In addition, the solubility of simple binary compounds such Eastern Samoa oxides and sulfides, both alcoholic bases, is often symbiotic along pH scale. In this section, we discuss the human relationship 'tween the solvability of these classes of compounds and pH.
The Effect of Acid–Base Equilibriums happening the Solubility of Salts
We begin our discussion by examining the effect of pH scale on the solubility of a representative salt, M+A−, where A− is the conjugate base of the weak sulphurous HA. When the salt dissolves in water, the following reaction occurs:
Equation 17.13
The anion can also react with water in a hydrolysis reaction:
Equation 17.14
Because of the chemical reaction described in Equation 17.14, the foretold solubility of a sparingly solvable SALT that has a basic anion such as S2−, PO4 3−, or CO3 2− is increased, as described in Section 17.1 "Determining the Solubility of Ionic Compounds". If instead a strong Lucy in the sky with diamonds is added to the solution, the added H+ will react essentially completely with A− to build HA. This reaction decreases [A−], which decreases the magnitude of the ion product (Q = [M+][A−]). According to Le Châtelier's principle, more Artium Magister will dissolve until Q = K sp. Hence an acidic pH dramatically increases the solvability of about complete sparingly soluble salts whose anion is the conjugate base of a weak acid. In line, pH has little to no more effect on the solvability of salts whose anion is the conjugate base of a stronger weak acid surgery a strong acid, respectively (e.g., chlorides, bromides, iodides, and sulfates). For example, the hydroxide salt Mg(OH)2 is relatively insolvable in pee:
Equation 17.15
When acid is added to a soaked resolution that contains excess solid Mg(OH)2, the succeeding reaction occurs, removing OH− from solution:
Equation 17.16
The overall equation for the reaction of Mg(OH)2 with acid is olibanum
Equality 17.17
A more acid is added to a abatement of Mg(Buckeye State)2, the equilibrium shown in Par 17.17 is driven to the right, so to a greater extent Mg(OH)2 dissolves.
Such pH-bloodsucking solvability is not restricted to salts that contain anions derived from water. For illustration, CaF2 is a sparingly soluble SALT:
Equating 17.18
When strong acid is added to a saturated resolution of CaF2, the following reaction occurs:
Equation 17.19
Because the forward reaction decreases the fluoride ion assiduousness, much CaF2 dissolves to relieve the stress on the scheme. The net profit response of CaF2 with strong acid is hence
Equivalence 17.20
CaF2(s) + 2H+(aq) → Ca2+(aq) + 2HF(aq)
Instance 7 shows how to calculate the solvability gist of adding a strong virulent to a solution of a sparingly soluble table salt.
Note the Pattern
Meagrely solvable salts derived from weak acids tend to be more soluble in an acid-forming solution.
Example 7
Lead oxalate (PbC2O4), lead iodide (PbI2), and lead sulfate (PbSO4) are all rather insoluble, with K sp values of 4.8 × 10−10, 9.8 × 10−9, and 2.53 × 10−8, severally. What effect does adding a strong virulent, such as perchloric acid, have on their congener solubilities?
Precondition: K sp values for three compounds
Asked for: relative solubilities in acid solution
Scheme:
Write the balanced chemical equation for the licentiousness of each salt. Because the strongest coupled root volition embody most affected by the addition of strong acid, mold the relative solubilities from the relative basicity of the anions.
Solution:
The solubility equilibriums for the triplet salts are as follows:
The addition of a strong caustic will have the greatest effect on the solubility of a salt that contains the conjugate base of a weak Lucy in the sky with diamonds atomic number 3 the anion. Because HI is a vehement acid, we predict that adding a intense acid to a saturated solution of PbI2 will not greatly affect its solubility; the bitter will only dissociate to form H+(aq) and the related to anion. In contrast, oxalate is the fully deprotonated form of ethanedioic acid (HO2CCO2H), which is a weak diprotic superman (pK a1 = 1.23 and pK a2 = 4.19). Consequently, the oxalate ion has a significant affinity for unmatchable proton and a let down kinship for a bit proton. Adding a strong acid to a concentrated solution of lead oxalate will resultant in the following reactions:
These reactions bequeath decrease [C2O4 2−], causation more than lead oxalate to dissolve to relieve the stress on the system.The pK a of HSO4 − (1.99) is similar in magnitude to the pK a1 of ethanedioic acid, so adding a strong acidic to a pure resolution of PbSO4 will outcome in the following reaction:
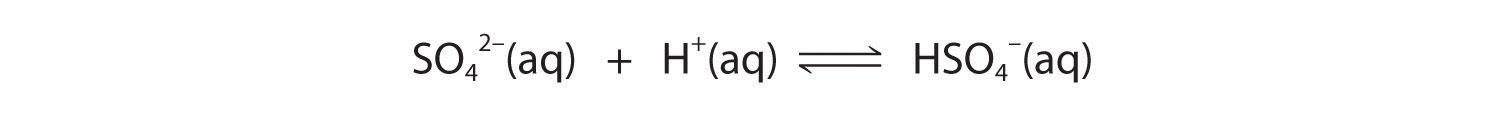
Because HSO4 − has a pKa of 1.99, this reaction leave lie largely to the left as written. Consequently, we predict that the effect of added substantial acid on the solubility of PbSO4 will embody significantly less than for PbC2O4.
Exercise
Which of the succeeding inexplicable salts—AgCl, Ag2CO3, Atomic number 473PO4, and/or AgBr—will be considerably more soluble in 1.0 M HNO3 than in complete piddle?
Answer: Ag2CO3 and Atomic number 473PO4
Caves and their associated pinnacles and spires of stone provide one of the most impressive examples of pH-dependent solubility equilibriums (set off (a) in Figure 17.6 "The Chemical science of Cave Constitution"). Perhaps the most familiar caves are formed from limestone, such as Carlsbad Caverns in New Mexico, Mammoth Cave in Kentucky, and Luray Caverns in Old Dominion. The primary reactions that are causative the formation of limestone caves are arsenic follows:
Equating 17.21
Equality 17.22
Equation 17.23
Figure 17.6 The Chemistry of Cave Formation

(a) This cave in Campanet, Mallorca, Kingdom of Spain, and its related formations are examples of pH-dependent solubility equilibriums. (b) A cave forms when groundwater containing part CO2, forming an acidic solution, dissolves limestone (CaCO3) in a process that may take tens of thousands of years. As groundwater seeps into a cave, water evaporates from the solution of CaCO3 in CO2-rich urine, producing a supersaturated root and a dislodge in equilibrium that causes precipitation of the CaCO3. The deposited limestone eventually forms stalactites and stalagmites.
Limestone deposits that form caves consist in the main of CaCO3 from the remains of surviving creatures such as clams and corals, which used it for making structures such arsenic shells. When a sodden solution of CaCO3 in CO2-rich H2O rises toward Ground's surface OR is other heated, CO2 gas is released as the water warms. CaCO3 then precipitates from the solution according to the following equation (part (b) in Figure 17.6 "The Chemistry of Cave Formation"):
Equation 17.24
The forward direction is the one reaction that produces the solid called scale in teapots, coffee makers, water heaters, boilers, and other places where thorny water is repeatedly heated.
When groundwater-containing atmospheric CO2 (Par 17.21 and Equation 17.22) finds its way into microscopic cracks in the limestone deposits, CaCO3 dissolves in the acidic solution in the overturn direction of Equivalence 17.24. The cracks step by step enlarge from 10–50 µm to 5–10 mm, a process that can take as longitudinal arsenic 10,000 yr. Eventually, after about another 10,000 yr, a spelunk forms. Groundwater from the surface seeps into the cave and clings to the cap, where the water evaporates and causes the equilibrium in Equivalence 17.24 to switching to the right. A circular layer of jellied CaCO3 is deposited, which eventually produces a long, hollow spire of limestone called a stalactite that grows down from the ceiling. Below, where the droplets land when they fall from the cap, a similar process causes some other steeple, titled a stalagmite, to mature up. The same processes that carve stunned hollows below ground are also at work above primer, in both cases producing fantastically convoluted landscapes like that of Yunnan Province in Communist China (Figure 17.7 "Solubility Equilibriums in the Establishment of Karst Landscapes").
Figure 17.7 Solubility Equilibriums in the Formation of Karst Landscapes

Landscapes such as the steep limestone pinnacles of the Stone Wood in Yunnan Province, PRC, are formed from the same litigate that produces caves and their associated formations.
Acidulent, Basic, and Amphoteric Oxides and Hydroxides
I of the earliest classifications of substances was supported their solubility in acidic versus basic solution, which led to the classification of oxides and hydroxides as being either base or acid. Basic oxidesAn oxide that reacts with water to acquire a BASIC solution or dissolves readily in liquid acid. and hydroxides either react with water to produce a basic solution or dissolve readily in aqueous acid. Acidic oxidesAn oxide that reacts with water supply to develop an acidic solution or dissolves in liquid base. Beaver State hydroxides either react with water to produce an acid solution or are soluble in binary compound base. As shown in Figure 17.8 "Classification of the Oxides of the Main Group Elements According to Their Acid-forming surgery Basic Character", there is a make correlation between the acidic operating room the basal fictitious character of an oxide and the set out of the element combined with oxygen in the rhythmic shelve. Oxides of metallic elements are generally basic oxides, and oxides of nonmetallic elements are acidulent oxides. Compare, for example, the reactions of a typical metal oxide, cesium oxide, and a characteristic nonmetal oxide, sulfur trioxide, with water:
Equation 17.25
Equating 17.26
Caesium oxide reacts with body of water to produce a basic solution of cesium hydroxide, whereas S trioxide reacts with water to produce a solution of element acid—very different behaviors indeed!
Note the Radiation pattern
Metal oxides generally react with water to produce basic solutions, whereas nonmetal oxides produce acidic solutions.
The difference in reactivity is ascribable the departure in bonding in the two kinds of oxides. Because of the deficient electronegativity of the metals at the far left in the periodic table, their oxides are best viewed as containing discrete Mn + cations and O2− anions. At the other end of the spectrum are metalloid oxides; due to their higher electronegativities, nonmetals form oxides with covalent bonds to atomic number 8. Because of the high electronegativity of oxygen, yet, the covalent bond between oxygen and the other mote, E, is usually polarized: Eδ+–Oδ−. The molecule E in these oxides acts as a Harry Sinclair Lewis loony toons that reacts with the O atom of water to produce an oxoacid. Oxides of metals in high oxidation states also tend to be acidic oxides for the same conclude: they carry covalent bonds to oxygen. An example of an acidulous metal-looking oxide is Low3, which is insoluble in both water and dose but dissolves in strong base to give solutions of the molybdate ion (MoO4 2−):
Equation 17.27
MoO3(s) + 2OH−(aq) → Low4 2−(aq) + H2O(l)
As shown in Figure 17.8 "Sorting of the Oxides of the Main Group Elements According to Their Acidic or Basic Character reference", there is a gradual transition from basic bronze oxides to acidic nonmetallic oxides as we go from the lower left to the upper right in the periodic table, with a broad diagonal band of oxides of average character separating the deuce extremes. Many of the oxides of the elements in this diagonal region of the periodic table are soluble in both acidic and basic solutions; consequently, they are called amphiprotic oxidesAn oxide that privy dissolve in acid to get water supply and unthaw in base to produce a soluble multifactorial. (from the Greek ampho, meaning "some," Eastern Samoa in amphoteric, which was defined in Chapter 16 "Aqueous Pane–Base Equilibriums", Section 16.1 "The Autoionization of Pee"). Amphoteric oxides either dissolve in acid to produce water supply Beaver State dissolve in base to produce a oil-soluble complex. As shown in Figure 17.9 "Chromium(III) Hydroxide [Cr(OH)", e.g., mixing the amphoteric oxide Cr(OH)3 (also written as Cr2O3·3H2O) with piss gives a sloughy, chromatic suspension. Adding acerbic causes the Cr(OH)3 to dissolve to give a bright violet solution of Chromium3+(aq), which contains the [Cr(H2O)6]3+ ion, whereas adding strong base gives a political party solution of the [Cr(Ohio)4]− ion. The chemical equations for the reactions are arsenic follows:
Equation 17.28
Equation 17.29
Soma 17.8 Assortment of the Oxides of the Intense Aggroup Elements According to Their Acidulous or Basic Character
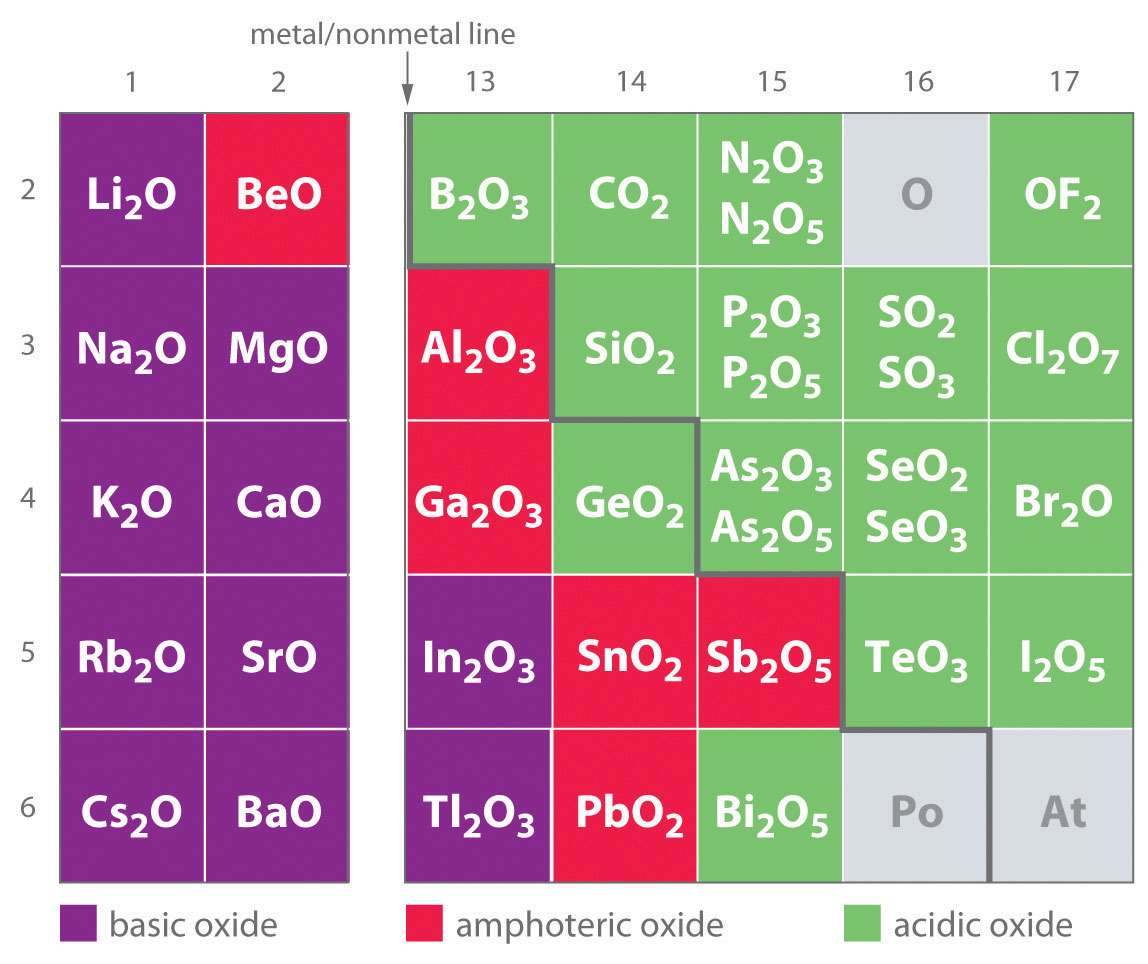
There is a gradual transition from radical oxides to acidic oxides from the lower left to the upper right-minded in the periodic table. Oxides of metallic elements are generally basic oxides, which either react with water to chassis a basic solution or dissolve in aqueous Lucy in the sky with diamonds. In contrast, oxides of nonmetallic elements are acid oxides, which either oppose with water to form an acidic answer OR are soluble in aqueous base. Oxides of intermediate character, called amphoteric oxides, are located along a diagonal delineate between the ii extremes. Amphiprotic oxides either dissolve in acid to produce weewe or dissolve in base to produce a fat-soluble complex ion. (Radioactive elements are not classified advertisement.)
Cipher 17.9 Chromium(III) Hydroxide [Cr(Ohio)3 Beaver State Cr2O3·3H2O] Is an Example of an Amphoteric Oxide

All three beakers originally contained a suspension of brownish purple Chromium(OH)3(s) (nerve centre). When concentrated acid (6 M H2SO4) was added to the beaker connected the left, Cr(OH)3 dissolved to get violet [Chromium(H2O)6]3+ ions and water. The addition of concentrated base (6 M NaOH) to the beaker on the mighty caused Cr(OH)3 to dissolve, producing Green River [Cr(Buckeye State)4]−ions.
Example 8
Aluminum hydroxide, written as either Al(OH)3 or Atomic number 132O3·3H2O, is amphoteric. Publish chemical equations to describe the profligacy of aluminum hydroxide in (a) acid and (b) base.
Given: amphoteric compound
Asked for: dissolution reactions in vitriolic and base
Strategy:
Using Equation 17.28 and Equation 17.29 as a guide, write the dissolution reactions in acid and base solutions.
Result:
-
An acid donates protons to hydroxide to give weewe and the hydrated metal ion, so aluminum hydroxide, which contains three OH− ions per Al, needs three H+ ions:
Aluminium(Buckeye State)3(s) + 3H+(aq) → Aluminium3+(aq) + 3H2O(l)In aqueous solution, Al3+ forms the Gordian ion [Aluminium(H2O)6]3+.
-
In basic solution, OH− is added to the compound to acquire a soluble and stable poly(hydroxo) complex:
Al(OH)3(s) + OH−(aq) → [Al(OH)4]−(aq)
Exercise
Bull(II) hydroxide, graphic as either Copper(OH)2 or CuO·H2O, is amphoteric. Write chemical equations that describe the dissolution of cupric hydrated oxide both in an blistering and in a base.
Answer:
Cu(OH)2(s) + 2H+(aq) → Cu2+(aq) + 2H2O(l) Cu(OH)2(s) + 2OH−(aq) → [Cu(OH)4]2−(aq)
Selective Precipitation Victimisation pH
Galore liquified metal ions can be separated by the selective precipitation of the cations from solution under particular conditions. In this technique, pH is frequently accustomed control the concentration of the anion in solution, which controls which cations precipitate.
Note the Pattern
The concentration of anions in solution can often make up controlled by adjusting the pH, thereby allowing the selective precipitation of cations.
Suppose, for exemplar, we have a solution that contains 1.0 mM Zn2+ and 1.0 millimeter Cd2+ and wishing to separate the two metals by discriminating precipitation as the insoluble sulfide salts, ZnS and CdS. The in dispute solubility equilibriums can be written A follows:
Equality 17.30
Equation 17.31
Because the S2− ion is rather introductory and reacts extensively with water to give Element 108− and OH−, the solubility equilibriums are more accurately written As kinda than Here we use the simpler signifier involving S2−, which is even because we consider the reaction of S2− with pee into invoice later in the solution, arriving at the same answer exploitation either equilibrium equation.
The sulfide concentrations needed to causal agent ZnS and CdS to precipitate are as follows:
Equivalence 17.32
Equation 17.33
Thus sulfide concentrations between 1.6 × 10−21 M and 8.0 × 10−24 M will precipitate CdS from solution but not ZnS. How do we obtain such low concentrations of sulfide? A saturated aqueous solution of H2S contains 0.10 M H2S at 20°C. The pK a1 for H2S is 6.97, and pK a2 corresponding to the formation of [S2−] is 12.90. The equations for these reactions are as follows:
Equivalence 17.34
We can show that the concentration of S2− is 1.3 × 10−13 by comparing K a1 and K a2 and recognizing that the contribution to [H+] from the dissociation of HS− is negligible compared with [H+] from the dissociation of H2S. Thus subbing 0.10 M in the par for K a1 for the assiduousness of H2S, which is essentially constant regardless of the pH, gives the following:
Equivalence 17.35
Substituting this value for [H+] and [Atomic number 108−] into the equation for K a2,
Although [S2−] in an H2S solution is very low (1.3 × 10−13 M), effervescent H2S through the solvent until it is saturated would precipitate both metal ions because the density of S2− would and then be much greater than 1.6 × 10−21 M. Thus we essential adjust [S2−] to stick around within the desired range. The about straight way to do this is to adjust [H+] by adding sulfurous to the H2S root (recall Le Châtelier's rule), thereby driving the chemical equilibrium in Equation 17.34 to the near. The boilers suit equation for the dissociation of H2S is as follows:
Equation 17.36
Now we dismiss use the equilibrium constant K for the overall reaction, which is the product of K a1 and K a2, and the density of H2S in a intense solvent to bet the H+ concentration needed to produce [S2−] of 1.6 × 10−21 M:
Par 17.37
Equation 17.38
Thus adding a strong Zen such as HCl to make the solution 0.94 M in H+ will keep the Thomas More soluble ZnS from precipitating while ensuring that the less soluble CdS will precipitate when the root is sopping with H2S.
Example 9
A solution contains 0.010 M Ca2+ and 0.010 M La3+. What concentration of HCl is needful to precipitate La2(C2O4)3·9H2O but not Calcium(C2O4)·H2O if the concentration of oxalic acid is 1.0 M? K sp values are 2.32 × 10−9 for Ca(C2O4) and 2.5 × 10−27 for Pelican State2(C2O4)3; pK a1 = 1.25 and pK a2 = 3.81 for ethanedioic acid.
Presented: concentrations of cations, K sp values, and concentration and pK a values for oxalic superman
Asked for: concentration of HCl needed for discriminating hurry of La2(C2O4)3
Scheme:
A Write each solubility product expression and calculate the oxalate concentration needed for precipitation to occur. Determine the concentration range needed for selective precipitation of La2(C2O4)3·9H2O.
B Add the equations for the first and second gear dissociations of ethanedioic acid to get an gross equating for the dissociation of oxalic acid to oxalate. Substitute the [ox2−] needed to hurried La2(C2O4)3·9H2O into the overall equation for the disassociation of ethanedioic acid to estimate the required [H+].
Solution:
A Because the salts have different stoichiometries, we cannot directly compare the magnitudes of the solubility products. Instead, we must utilize the equilibrium constant expression for each solubility product to calculate the concentration of oxalate required for precipitation to happen. Using ox2− for oxalate, we write the solubility product expression for calcium oxalate as follows:
The expression for lanthanum oxalate is as follows:
Thus La oxalate is less soluble and will selectively precipitate when the oxalate concentration is betwixt 2.9 × 10−8 M and 2.32 × 10−7 M.
B To prevent Atomic number 202+ from precipitating as atomic number 20 oxalate, we must add sufficient H+ to consecrate a maximum oxalate concentration of 2.32 × 10−7 M. We backside calculate the required [H+] by victimization the total equality for the dissociation of oxalic acid to oxalate:
Substituting the desired oxalate assiduity into the equilibrium constant reflexion,
Thusly adding enough HCl to give [H+] = 6.1 M will cause single La2(C2O4)3·9H2O to hurried from the answer.
Exercise
A solution contains 0.015 M Fe2+ and 0.015 M Pb2+. What concentration of acid is needed to ensure that Pb2+ precipitates as PbS in a saturated solution of H2S, but Fe2+ does not precipitate as FeS? K sp values are 6.3 × 10−18 for FeS and 8.0 × 10−28 for PBS.
Solution: 0.018 M H+
Summary
The anion in many sparingly soluble salts is the conjugate base of a weak acid. At miserable pH, protonation of the anion can dramatically increase the solubility of the salt. Oxides can exist classified as acidulent oxides surgery basic oxides. Acidulent oxides either react with water to give an acidic solution Oregon dissolve in sinewy cornerstone; most acidic oxides are nonmetal oxides or oxides of metals in high oxidization states. Elemental oxides either oppose with body of water to give way a base solution or dissolve in noticeable acid; most basic oxides are oxides of metallic elements. Oxides or hydroxides that are water-soluble in both acidic and basic solutions are called amphoteric oxides. Most elements whose oxides exhibit amphoteric behavior are located along the diagonal lineage separating metals and nonmetals in the periodic mesa. In solutions that contain mixtures of dissolved metal ions, the pH can be accustomed control the anion concentration needed to by selection precipitate the desirable cation.
Key Takeout food
- The anion in sparingly soluble salts is often the conjugate base of a gutless acid that May get over protonated in solution, thusly the solubility of simple oxides and sulfides, both strong bases, often depends on pH.
Conceptual Problems
-
Which of the following will show the greatest gain in solvability if 1 M HNO3 is used instead of distilled irrigate? Explain your reasoning.
- CuCl2
- K[Pb(OH)3]
- Ba(CH3Atomic number 272)2
- CaCO3
-
Of the compounds Sn(CH3CO2)2 and SnS, one is soluble in dilute HCl and the other is soluble solely in hot, piled HCl. Which is which? Provide a reasonable account.
-
Where in the periodic table do you have a bun in the oven to find elements that form basic oxides? Where do you anticipate to find elements that form acidic oxides?
-
Because water can autoionize, it reacts with oxides either A a base (as OH−) or every bit an acid (as H3O+). Make you wait oxides of elements in high oxidisation states to be more acidic (reacting with OH−) or more basic (reacting with H3O+) than the corresponding oxides in low oxidation states? Why?
-
Given concrete samples of CRO, Cr2O3, and CrO3, which would you expect to be the most acidic (reacts most readily with Ohio−)? Which would embody the most basic (reacts most promptly with H3O+)? Why?
-
Which of these elements—Exist, B, Al, N, Se, In, Tl, Pb—do you expect to form an amphoteric oxide? Why?
Definite quantity Problems
-
A 1.0 L solution contains 1.98 M Atomic number 13(NO3)3. What are [OH−] and [H+]? What pH is required to come down the cation as Al(OH)3? K sp = 1.3 × 10−33 and K a = 1.05 × 10−5 for the hydrous Atomic number 133+ ion.
-
A 1.0 L solution contains 2.03 M CoCl2. What is [H+]? What pH is requisite to precipitous the cation as Co(Buckeye State)2? K sp = 5.92 × 10−15 and K a = 1.26 × 10−9 for the hydrated Co2+ ion.
-
Given 100 mL of a solution that contains 0.80 millimeter Ag+ and 0.80 mM Cu+, tin the two metals exist injured by discriminating precipitation as the insolvable bromide salts by adding 10 mL of an 8.0 mM solution of KBr? K sp values are 6.27 × 10−9 for CuBr and 5.35 × 10−13 for AgBr. What maximum [Br−] testament separate the ions?
-
Given 100 mL of a resolution that is 1.5 mM in Tl+, Zn2+, and Ni2+, which ions can be detached from resolution by adding 5.0 cubic centimetre of a 12.0 mM solution of Na2C2O4?
Precipitate K sp Tl2C2O4 2 × 10−4 ZnC2O4·2H2O 1.38 × 10−9 NiC2O4 4 × 10−10 How many milliliters of 12.0 mM Na2C2O4 should be added to separate Thallium+ and Zn2+ from Ni2+?
Answers
-
[H+] = 4.56 × 10−3; [OH−] = 2.19 × 10−12; pH = 2.94
-
No; both metal ions will precipitant; AgBr will precipitate as Br− is added, and CuBr will begin to precipitate at [Bromine−] = 8.6 × 10−6 M.
calculate the solubility of mg oh 2 in water
Source: https://saylordotorg.github.io/text_general-chemistry-principles-patterns-and-applications-v1.0/s21-04-solubility-and-ph.html
Posting Komentar untuk "calculate the solubility of mg oh 2 in water"